Abstract
Introduction
Immunoglobulin M monoclonal gammopathy of undetermined significance (IgM MGUS) is a relatively common pre-malignant condition that may progress to Waldenstrom macroglobulinemia (WM), an incurable low-grade non-Hodgkin lymphoma. The acquisition of genomic alterations in a multistep process of progression is likely a central mechanism responsible for the progression of IgM MGUS to WM, leading to increased tumor burden and end-organ damage. The exact role of the immune system in this process is still poorly characterized. The modulation of the immune system by malignant cells leading to anti-lymphoma immunity or immune evasion is a well-stablished principle associated with lymphoma survival and progression, respectively. The understanding of the pathobiology of IgM MGUS and WM has grown significantly over the last few years; however, most of the past research has focused on genetics and flow cytometric analysis of malignant cells. The introduction of CyTOF, a novel platform coupling mass spectometry with single-cell flow cytometry using antibody-metal isotope pairs, has allowed a broader single-cell analysis and the study of functional profiles. We here report the use of CyTOF technology to comprehensively profile the immune system of patients with IgM MGUS and WM.
Methods
Viably-cryopreserved (DMSO 10%) single-cell suspensions containing peripheral blood mononuclear cells from 15 patients (WM n=9 and IgM MGUS n=6) were used in this study. Recovered cells (1-3 x 106 per sample) were stained with a 30-parameter surface protein CyTOF panel designed to characterize multiple cell types and profile the immune system. Nucleated cellular events were identified using a DNA intercalator conjugated to natural abundance iridium (191Ir and 193Ir). Cisplatin (195Pt) was used for dead-live cell discrimination and calibration beads containing natural abundance cerium (140/142Ce), europium (151/153Eu), holmium (165Ho), and lutetium (175/176Lu) were used for normalization of the instrument signal. CyTOF analysis of normalized files was carried out using the Astrolabe Diagnostics platform. Cell clusters were assigned using the FlowSOM package and differential abundance analysis was performed using the edgeR package. Analysis is adjusted for multiple comparisons.
Results
The median live single-cell count for the entire cohort was 573,300 (range 50,681 to 869,727), representing a median of 81% (range 39% to 87%) of the total event counts [median 820,800 (range 130,452 to 1,000,000]. The cell count and the proportion of event count was similar in patients with WM or IgM MGUS. T cells represented 45.8% of the total events; granulocytes represented 31.3%; NK cells represented 8.6%; monocytes represented 8.1%; B cells represented 2.4% of the total events for the entire cohort.
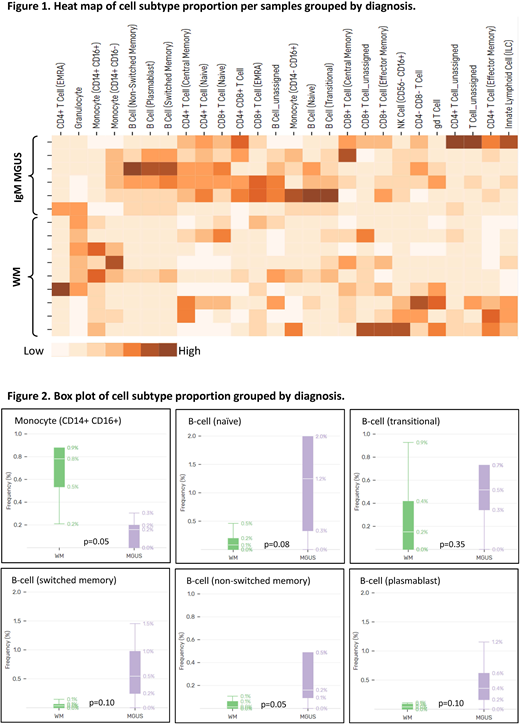
Figure 1 shows a heat map of different immune cell sub-types proportion per patient sample. A difference was noted in the proportion of monocytes (CD14+, CD16+) and B-cells when comparing patients with IgM MGUS and WM (figure 2). Patients with WM had a higher proportion of monocytes compared to IgM MGUS patients (p=0.05). A higher proportion of B-cells was noted in patients with IgM MGUS when compared to WM (4.2% vs 0.6%, p=0.01). When B-cells were sub-classified as naïve, transitional, non-switched memory, switched memory, and plasmablasts, the proportion of non-switched memory B-cells was higher in IgM MGUS compared to WM (p=0.05). While the results of this pilot study suggest important differences between IgM MGUS and WM as regards the immune repertoire, confirmatory studies are in progress in a larger cohort of patients.
Conclusion
To our knowledge, this is the first study of mass cytometry in patients with WM and IgM MGUS patients. A higher proportion of monocytes and lower proportion of memory B-cells was noted in patients with WM compared to IgM MGUS. CyTOF study of the immune profile in these patients is feasible and can potentially uncover relationships between cell types not typically associated with the disease progression continuum (IgM MGUS to WM). While definitive conclusion cannot be made using this dataset due to the small sample size, CyTOF is a powerful tool in the study of the immune profile of WM patients.
Gertz:Amgen: Consultancy; Abbvie: Consultancy; Apellis: Consultancy; annexon: Consultancy; spectrum: Consultancy, Honoraria; Teva: Consultancy; Alnylam: Honoraria; Physicians Education Resource: Consultancy; Ionis: Honoraria; janssen: Consultancy; celgene: Consultancy; Prothena: Honoraria; Research to Practice: Consultancy; Medscape: Consultancy. Kapoor:Celgene: Research Funding; Takeda: Research Funding. Ailawadhi:Takeda: Consultancy; Amgen: Consultancy; Janssen: Consultancy; Celgene: Consultancy; Pharmacyclics: Research Funding. Reeder:Affimed: Research Funding. Ansell:Seattle Genetics: Research Funding; Regeneron: Research Funding; Takeda: Research Funding; Pfizer: Research Funding; Merck & Co: Research Funding; Trillium: Research Funding; Affimed: Research Funding; LAM Therapeutics: Research Funding; Celldex: Research Funding; Bristol-Myers Squibb: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal